E-mail:info@tradomed-invest.ru
Качественное офтальмологическое оборудование для профессионалов!
Тел.:
+7 495 662 7866
Трейдомед инвест / Страничка клинициста / IRIS Medical G-Probe
Страничка клинициста
IRIS Medical G-Probe
 Список
статей Список
статейНаконечник G-Probe>>> |
|
Laser System OcuLight
SL, OcuLight SLx, Compatibility: IQ 810 Treatment Beam: Infrared diode laser (810 nm) Aiming Beam: Red diode laser (650 nm) Spot Size: 550 micron at fiber tip 400 micron at ciliary body Fiber Length: 6’ (1.8 m) Handle Length: 2.5” (6.5 cm) The G-Probe is intended for single use only.  “
Cyclodiode is an effective treatment for refractory glaucoma of all etiologies.” “
Cyclodiode is an effective treatment for refractory glaucoma of all etiologies.”Murphy CC, Burnett CAM, Spry PGD, Broadway DC, Diamond JP. A two centre study of the dose-response relation for transscleral diode laser cyclophotocoagulation in refractory glaucoma. Br J Ophthalmol 87:1252-1257, 2003. 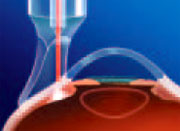 A side view showing the edge of the G-Probe placed on the surgical limbus.
The laser emitting fiber is placed directly over the pars plicata to ensure
delivery of the beam through the sclera and into the ciliary body.
A side view showing the edge of the G-Probe placed on the surgical limbus.
The laser emitting fiber is placed directly over the pars plicata to ensure
delivery of the beam through the sclera and into the ciliary body.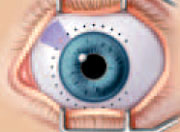 The design of the probe tip allows for precise placement around the circumference
of the limbus.
The design of the probe tip allows for precise placement around the circumference
of the limbus. A posterior view of laser treatment applied to the ciliary body. Eyes
are treated up to 3/4 of the circumference with approximately 18 applications
to effectively reduce IOP.
A posterior view of laser treatment applied to the ciliary body. Eyes
are treated up to 3/4 of the circumference with approximately 18 applications
to effectively reduce IOP. |
|
| G-Probe Product Benefits Include: Patented tip design offers consistent treatment and ease of use while patient is in a supine position.  The IRIS Medical G-Probe is used for transscleral cyclophotocoagulation
(TSCPC) of the ciliary processes. TSCPC has been shown to be a safe and
highly effective method for lowering intraocular pressure (IOP) in patients
with uncontrolled glaucoma.1-4 The G-Probe is used with IRIDEX
infrared laser systems, such as the OcuLight® SL/SLx and IQ 810™, which
provide an 810 nm treatment beam. The 810 nm wavelength easily passes through
the sclera without altering its permeability or ultrastructure,5,6
and is highly absorbed by the ciliary body pigment epithelium. TSCPC offers
a simple, accurate, effective and gentle treatment alternative to cyclocryotherapy
and Nd:YAG. Compared to cyclocryotherapy, it offers a higher rate of success
with less inflammation,7 fewer complications, shorter surgery
time, and the opportunity for retreatments.8 Compared to Nd:YAG,
it has a higher initial and repeat success rate,9 offers increased
patient comfort levels due to decreased inflammation, and provides a more
durable IOP reduction.10,11 TSCPC with the G-Probe is an efficient
and repeatable treatment that can easily be performed in your office.
The IRIS Medical G-Probe is used for transscleral cyclophotocoagulation
(TSCPC) of the ciliary processes. TSCPC has been shown to be a safe and
highly effective method for lowering intraocular pressure (IOP) in patients
with uncontrolled glaucoma.1-4 The G-Probe is used with IRIDEX
infrared laser systems, such as the OcuLight® SL/SLx and IQ 810™, which
provide an 810 nm treatment beam. The 810 nm wavelength easily passes through
the sclera without altering its permeability or ultrastructure,5,6
and is highly absorbed by the ciliary body pigment epithelium. TSCPC offers
a simple, accurate, effective and gentle treatment alternative to cyclocryotherapy
and Nd:YAG. Compared to cyclocryotherapy, it offers a higher rate of success
with less inflammation,7 fewer complications, shorter surgery
time, and the opportunity for retreatments.8 Compared to Nd:YAG,
it has a higher initial and repeat success rate,9 offers increased
patient comfort levels due to decreased inflammation, and provides a more
durable IOP reduction.10,11 TSCPC with the G-Probe is an efficient
and repeatable treatment that can easily be performed in your office. |
|
1. Kosoko O, Gaasterland D, Pollack I, Enger C, The Diode Laser Ciliary Ablation Study Group: Abrams D, Belcher CD, Gaasterland D, Liebmann J, Pollack I, Ritch R, Schuman J, Shields MB, Wise J. Long-term outcome of initial ciliary ablation with contact diode laser transscleral cyclophotocoagulation for severe glaucoma. Ophthalmology 1996;103:1294-1302. 2. Schlote T, Derse M, Rassmann K, Nicaeus T, Dietz K, Thiel HJ. Efficacy and safety of contact transscleral diode laser cyclophotocoagulation for advanced glaucoma. J Glaucoma 2001; 10:294-301. 3. Hauber FA, Scherer WJ. Influence of total energy delivery on success rate after contact diode laser transscleral cyclophotocoagulation: a retrospective case review and meta-analysis. J Glaucoma 2002;11:329-333. 4. Lirio JPM, Martinez JA, Altuna J, League J, Reyes RD. Single-setting treatment protocol for diode-laser transscleral cyclophotocoagulation. Philipp J Ophthalmol 2004;29(4):171-174. 5. Olsen T, Edelhauser H, Lim J, Geroski D: Human scleral permeability. Effects of age, cryotherapy, transscleral diode laser, and surgical thinning. Invest Ophthalmol Vis Sci 1995:36;1893-1903. 6. Palmer DJ, Cohen J,Torczynski E, Deutsch TA:Transscleral diode laser cyclophotocoagulation on autopsy eyes with abnormally thinned sclera. Ophthalmic Surg Lasers 1997:28;495-500. 7. Schlote T, Derse M, Zierhut M: Transscleral diode laser cyclophotocoagulation for the treatment of refractory glaucoma secondary to inflammatory eye diseases. Br J Ophthalmol 2000:84;999-1003. 8. Mistlberger A,Tschiderer H, Ruckhofer J, Grabner G: Transscleral contact diode laser cyclophotocoagulation for refractory glaucoma. Spektrum der Augenheilk-kunde 1997:11(2);45-48. 9. Klein S, Rockwood EJ, Baerveldt G: Diode laser versus contact transscleral Nd:YAG cyclophotocoagulation (CPC) for uncontrolled glaucoma. [ARVO Abstract] Invest Ophthalmol Vis Sci 1996 37(3):S261 Abstract nr 1192. 10.Campbell K: Cyclophotocoagulation: A comparative study using two different wavelengths, Nd:YAG (1064 nm) and diode (810 nm). Abstract.American Society for Laser Medicine and Surgery Lasers Surg Med 1996,8(suppl):94. Grant Laser Center, Columbus, OH. 11.Zweifach E,Wilensky J, Hillman I, Quinones R, Gemperli A: A prospective ramdomized trial of diode and Nd:YAG lasers in treating glaucoma. [ARVO Abstract] Invest Ophthalmol Vis Sci 1997 38(4):S170 Abstract Nr 844. |
|
 |
|



